Clinical Research Resources at the Sarah W. Stedman Nutrition and Metabolism Center and Aesthetics Building at the Duke Center for Living campus
The Duke Molecular Physiology Institute mission includes clinical research conducted in the Sarah W. Stedman Nutrition and Metabolism Center and the Aesthetics Center, both located at the Duke Center for Living (CFL) campus. Research at the Stedman Center focuses on human research on cardiovascular, endocrine, renal, and metabolic diseases. The facility links DMPI and other Duke investigators in several thematic clusters, including primarily a nutrition group, an incretin hormone group, and a group focusing on the molecular circuits underlying health benefits of physical activity. Each of these groups connect to multiple other scientists across the Duke School of Medicine, as illustrated in the three diagrams below.
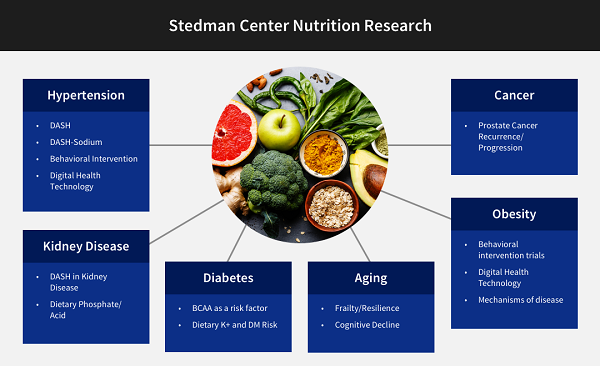
Contact for Nutrition Research collaborations: Laura Svetkey
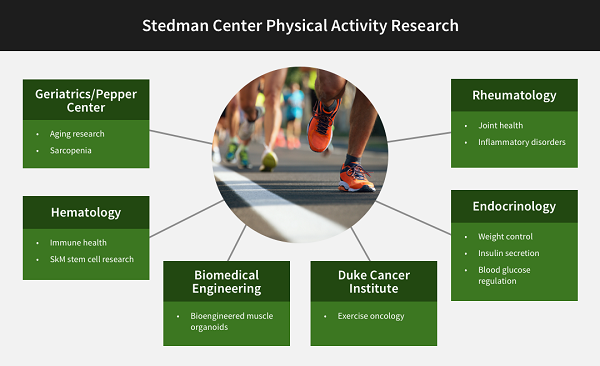
Contact for Physical Activity collaborations: William Kraus
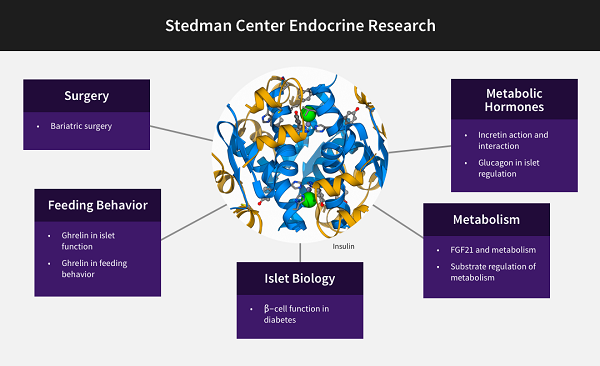
Contact for Endocrine Research collaborations:David D'Alessio
Resources for human physiology research at the Stedman Center
- Metabolic kitchen for controlled feeding studies: The Stedman Center Clinical Research building is a rare facility at Duke capable of delivering a controlled feeding intervention. In these studies, the nutrient content of food is predetermined and then chemically verified; all foods are prepared according to strict operating procedures in a metabolic kitchen. Study participants are provided with all study-specific food and beverages consumed either on site at the CFL campus or “to go” as pre-packaged meals for longer term dietary interventions.
- Space and expertise for behavioral intervention trials: The Stedman Center resources support rigorous methodology for group and individual coaching, cooking demonstrations, physical activity training, and collection of physiological, clinical, dietary, physical activity, and psychosocial data.
- Clinical exam unit: Four exam rooms, phlebotomy and procedure suite, lab-processing area, and the space and equipment for relevant clinical measurements (e.g., weight, blood pressure; both automatic and 24-hour ambulatory).
- Dietary assessment: The collection of dietary intake information using various methodologies including food recall, dietary record and food frequency questionnaires (paper and on-line).
- Cardiopulmonary exercise (CPX) test: A non-invasive exercise test to assess fitness level and cardiovascular response to exercise stress via ECG stress system monitoring and expired gas analysis.
- Resting metabolic rate (RMR) test: A test to measure the amount of energy the body uses while at rest via the use of a metabolic cart.
- Oral and intravenous glucose tolerance testing, and clamp and infusion studies: These methods provide robust measures of glucose tolerance, insulin sensitivity, and substrate metabolism in humans.
- Metabolic flux analysis using stable isotopes: Via infusion of stable isotope-labeled nutrients such as 13C glucose or 15N valine, we can follow their utilization through various metabolic pathways under different physiologic conditions.
- Assessment of insulin secretion: Hyperglycemic clamps, incretin and amino acid infusion and graded glucose infusions to measure insulin secretion in diabetic and nondiabetic subjects.
- BODPOD body composition: An evaluation of fat mass, lean body mass and % body fat using a BODPOD® chamber that uses air displacement to measure body volume.
- Muscle biopsy services: An outpatient surgical procedure to remove small tissue samples under local anesthetic. These muscle biopsy services provide tissue for multiple types of assays including muscle histology, protein, DNA, transcriptomic and metabolomic analyses.
- eHealth and mHealth technology innovations: The group deploys various wearable devices for monitoring physical activity (e.g. Fitbit, Actigraph, Garmin trackers). In addition, several CFL investigators and collaborators (e.g., Svetkey, Bosworth Bennett, Steinberg) investigate the use of wearable, hand-held, phone and on-line technology for dietary intervention, physical activity and weight control.
Ideal facility for clinical trial execution: The CFL provides Duke a unique resource for clinical trial execution by providing the core services listed above in a geographic location that is near Duke Hospital and our investigative teams, but at the same time, off-site at a more easily accessible location for our study participants.
